Abstract
Activating mutations of FLT3 (FLT3MT) gene are one of the most frequent pathogenic lesions in AML. As a consequence, tailored therapeutics have been developed in this setting. For instance, protein tyrosine kinase (PTK) domain is a target of inhibitors, which have already entered clinical practice. Overall, FLT3MT occur in about 30% of AML cases, with the internal tandem duplication (ITD) being by far the most common mutation. Indeed, FLT3ITD account for about 83% of all FLT3MT cases and are associated with dismal prognosis. In addition to FLT3ITD, distinct point mutations and deletions within the tyrosine kinase domain (FLT3TKD) and more specifically its activation loop (FLT3AL) have been described and can be also targeted by PTK inhibitors. Nonetheless, these latter mutations are less common, making up approximately 7-10% of all AML cases. Besides these "canonical” mutation types, point mutations outside of the FLT3AL, or "non-canonical” FLT3MT (FLT3NC) have also been observed in AML and other related myeloid neoplasms (MN). However, the prognostic impact and clinical relevance of such molecular alterations are not fully elucidated.
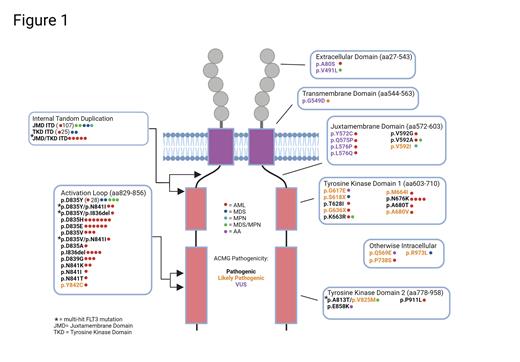
This study was inspired by a case diagnosed with CMML harboring a FLT3NC mutation at the amino acid position 592 substituting an alanine for a valine. This spot is immediately located at the starting of the region (591-597) and is important for maintaining the normal kinase in an inactive state in absence of the ligand. Of note is that treatment with the highly-selective, second generation PTK inhibitor, gilteritinib resulted in a good clinical response.
Therefore, we reviewed NGS sequencing results of 5,308 patients with MN in search for cases carrying similar lesions. A total of 248 patients with somatic FLT3MT were identified with 152 FLT3ITD, 66 FLT3AL, and 30 FLT3NC hits (Fig.1). The ITD subgroup included 144 AML, 4 MDS, 2 MPN, and 2 MDS/MPN overlap cases. Within cases with FLT3AL mutations, diagnoses consisted of 61 AML, 2 MDS, and 3 MDS/MPN. Most importantly, we also found 30 cases with FLT3NC mutations including 19 AML, 3 MDS, 1 MPN, 4 MDS/MPN, and notably 3 MDS cases with an antecedent history of aplastic anemia. While all the FLT3AL were predicted to be pathogenic based on the ACMG criteria, the FLT3NC consisted of 7 VUS, 10 likely pathogenic, and 9 known pathogenic variants with one biallelic, two recurrent variants in 2 patients and one (N676K) recurring in 4 patients. These atypical FLT3MT mapping to non-canonical locations were found more commonly in non-AML entities (37% in MDS/MPN, MDS, MPN vs. 63% AML) vs.FLT3AL (92% in AML; P< .001) and FLT3ITD (95% AML; P<.001).
Co-mutational burden was evaluated with an NGS panel of 51 commonly mutated genes in MN. As expected, NPM1MT was significantly more likely to be associated with cases carrying FLT3ITD (46%) than FLT3NC (20%) (P= .008). Additionally, DNMT3A was found to be recurrently associated to cases with both FLT3ITD (36%) and FLT3AL (30%) as compared to FLT3NC (10%) (P= .005, P= .023, respectively). Median VAFs of both FLT3ITD and FLT3AL were not different compared to FLT3NC (21 vs. 31%; P= .08).
Analysis of clinical phenotypes revealed that FLT3AL was more associated with high-grade diseases compared to FLT3NC (94% vs. 67%; P= .001). However, there was no difference in overall survival (OS) between FLT3AL and FLT3NC in AML (23 vs. 36 months; P= .70). In terms of treatment, 17 patients: 12 FLT3AL and 5 FLT3NC (4 AML, 1 MDS/MPN) received TKI inhibitors. In this limited group, no OS differences between AML patients treated with PTK inhibitors (13 cases receiving midostaurin, 3 gilteritinib and 1 sorafenib) and non-treated patients were observed in either FLT3AL or FLT3NC. However, all 5 patients harboring FTL3NC responded to PTK inhibitors.
In sum, the well-studied FLT3ITD and NPM1 association does not extend to point mutations outside the FLT3AL. In addition, although differences were noted in less advanced diseases, once patients progressed to AML no specific features, clinical parameters or prognosis differed between cases with FLT3 point mutations/deletions within the activation loop, which appeared to be similar to patients with other types of point mutations outside this region. We also report on the use of PTKs in FLT3NC, whose therapeutic response in association with such FLT3 molecular patterns should warrant further studies.
Disclosures
Carraway:CTI Biopharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy, Honoraria, Speakers Bureau; Jazz: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Stemline: Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Other: DSMB; Syndax: Other: DSMB; Takeda: Other: DSMB. Mukherjee:Celgene/Acceleron: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Advisor or review panel participant, Research Funding; Genentech: Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Advisor or review panel participant, Research Funding; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Advisor or review panel participant, Research Funding; Blueprint Medicines: Membership on an entity's Board of Directors or advisory committees, Other: Advisor or review panel participant; Eusa Pharma: Consultancy, Other: Advisor or review panel participant; Teaching and Speaking; Partnership for Health Analytic Research, LLC: Honoraria; McGraw Hill Hematology Oncology Board Review: Honoraria, Other: Advisor or review panel participant; Aplastic Anemia and MDS International Foundation: Honoraria; BioPharm: Consultancy; Jazz Pharmaceuticals: Other: Principal investigator for Investigator Initiated Trials (the Institution gets the funding), Research Funding; AbbVie: Membership on an entity's Board of Directors or advisory committees, Other: Advisor or review panel participant. Gerds:Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees, Research Funding; Imago BioSciences: Research Funding; Kratos Pharmaceuticals: Research Funding; Accurate Pharmaceuticals: Research Funding; Bristol Myers Squibb/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Incyte Corporation: Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sierra Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; Morphosys/Constellation: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; PharmaEssentia: Consultancy, Membership on an entity's Board of Directors or advisory committees. Sekeres:Bristol Myers-Squibb: Membership on an entity's Board of Directors or advisory committees; Takeda/Millenium: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Kurome: Membership on an entity's Board of Directors or advisory committees. Maciejewski:Alexion: Consultancy; Apellis Pharmaceuticals: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal